U.S. National WCD Experience
<< Back to all published studies
Aggregate National Experience With the Wearable Cardioverter Defibrillator: Event Rates, Compliance, and Survival
Purpose
- This study was created to review efficacy, wear time, and long-term survival for patients who wore the LifeVest Wearable Cardioverter Defibrillator (WCD) compared to patients who received an ICD.
Methods
- Retrospective analysis of U.S. patients (n=3,569) with a mean age of 59.3± 14.7 years that were recorded in a nationwide registry of post-market release WCDs.
- Includes all patients who wore the LifeVest WCD at some point between August 2002 and December 2006.
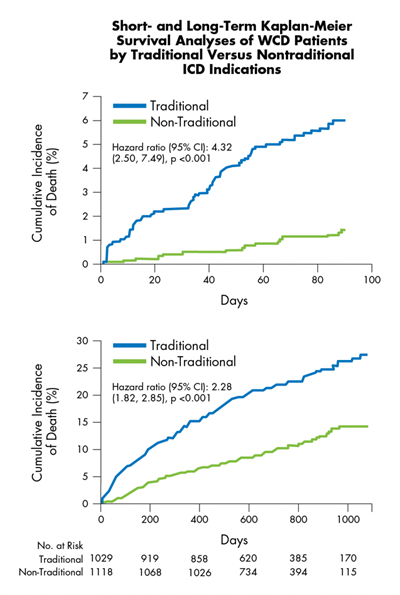
- Survival, using the Social Security Death Index (SSDI), was compared to the survival of Cleveland Clinic patients who underwent ICD implantation.
Key Results
- 80 sustained VT/VF events occurred in 59 patients (1.7%). First shock success was 76 of 76 (100%) for unconscious VT/VF and 79 of 80 (99%) for all VT/VF.
- Daily use of WCD was >90% in 52% of patients and >80% in 71% of patients. Median daily use was 21.7 hours or 91% of time available. Longer duration of monitoring correlated with higher wear time rates.
- During WCD use, overall acute survival was 99.2%. Long-term mortality rates in ICD and WCD groups were not significantly different for 3 years and 3 months timeframe.
Conclusions
- The WCD is highly effective in converting VT/VF on the first shock.
- Patient wear time of the WCD is high. Daily use of the WCD was >90% by most patients.
- Long-term survival data indicates WCD therapy is comparable to ICD therapy, rationalizing use of the WCD as a bridge to long-term ICD implantation.
- The WCD is a reasonable treatment option for patients with uncertain, long-term status awaiting a permanent ICD.
Click here to read the full publication
Chung MK, Szymkiewicz SJ, Shao M, et al. Aggregate national experience with the wearable cardioverter-defibrillator: Event rates, compliance, and survival. J Am Coll Cardiol. 2010;56(3):194–203.